How does x-ray crystallography of macromolecules work?
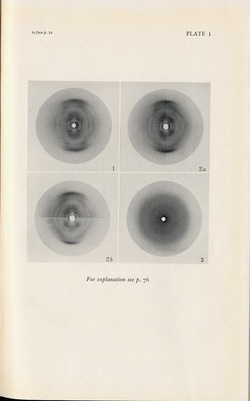 1947 DNA X-ray diffraction patterns
1947 DNA X-ray diffraction patterns
Creating the Crystal
Whatever the material to be examined, for X-ray crystallography the substance must be crystallized first, because the symmetric nature of crystals is ideal for interpreting the diffraction patterns. Creating the crystal involves isolation of the target molecule, generally a protein molecule. This can be tricky and a challenge for the chemists because each protein structure is unique, however certain methods have proven effective. Also, the use of cryogenics when the experiments are being performed help to "freeze" the sample in place.
Controlling the X-rays
The basics of this precise technique in almost all forms is the use of an X-ray beam, which passes through a prepared crystal sample. The sample is placed in the path of the X-rays, generally on a goniometer, which rotates around an axis (see animation below) for complete exposure to the X-ray. This allows the entire sample to be examined so that a structure can be more clearly determined. Before sophisticated and specific technology for controlling and refining the X-rays was created, the rays were less bright, had less flux, and therefore were much harder to use for a clear image. Long exposure times meant a great margin for error, which now can be eliminated.
Detectors
Films was the original detector of the X-ray scattering patterns, which was not only tedious to work with, but extremely time-consuming to develop and then analyze by hand. It required a long exposure time and could be foggy. The image was more difficult to see clearly because of this, and the low dynamic range meant that there was little contrast between the dark and light. Together, these factors pushed scientists to create image plates. These plates were coated in a material that would pick up the incoming X-rays and record where they hit. Much faster than films, these plates also had a greater dynamic range, so interpreting the data was more intuitive. Additionally, the plates were reusable after being washed, and could work well under a high energy beam.
Data Analysis
Prior to the widespread use of computer and software for the processing of data, the only way to truly understand the findings of X-ray crystallography was by carefully examining the diffraction patterns with the naked eye, then interpreting the findings. There were charts of brightness that the patterns could be compared to, and the phasing had to be done by humans as well. Additionally, without the large databases of protein and other macromolecule structures that are common today, there was little to compare against. This meant that nearly every new molecule had to be solved completely, and no molecular replacement could occur.
Whatever the material to be examined, for X-ray crystallography the substance must be crystallized first, because the symmetric nature of crystals is ideal for interpreting the diffraction patterns. Creating the crystal involves isolation of the target molecule, generally a protein molecule. This can be tricky and a challenge for the chemists because each protein structure is unique, however certain methods have proven effective. Also, the use of cryogenics when the experiments are being performed help to "freeze" the sample in place.
Controlling the X-rays
The basics of this precise technique in almost all forms is the use of an X-ray beam, which passes through a prepared crystal sample. The sample is placed in the path of the X-rays, generally on a goniometer, which rotates around an axis (see animation below) for complete exposure to the X-ray. This allows the entire sample to be examined so that a structure can be more clearly determined. Before sophisticated and specific technology for controlling and refining the X-rays was created, the rays were less bright, had less flux, and therefore were much harder to use for a clear image. Long exposure times meant a great margin for error, which now can be eliminated.
Detectors
Films was the original detector of the X-ray scattering patterns, which was not only tedious to work with, but extremely time-consuming to develop and then analyze by hand. It required a long exposure time and could be foggy. The image was more difficult to see clearly because of this, and the low dynamic range meant that there was little contrast between the dark and light. Together, these factors pushed scientists to create image plates. These plates were coated in a material that would pick up the incoming X-rays and record where they hit. Much faster than films, these plates also had a greater dynamic range, so interpreting the data was more intuitive. Additionally, the plates were reusable after being washed, and could work well under a high energy beam.
Data Analysis
Prior to the widespread use of computer and software for the processing of data, the only way to truly understand the findings of X-ray crystallography was by carefully examining the diffraction patterns with the naked eye, then interpreting the findings. There were charts of brightness that the patterns could be compared to, and the phasing had to be done by humans as well. Additionally, without the large databases of protein and other macromolecule structures that are common today, there was little to compare against. This meant that nearly every new molecule had to be solved completely, and no molecular replacement could occur.
