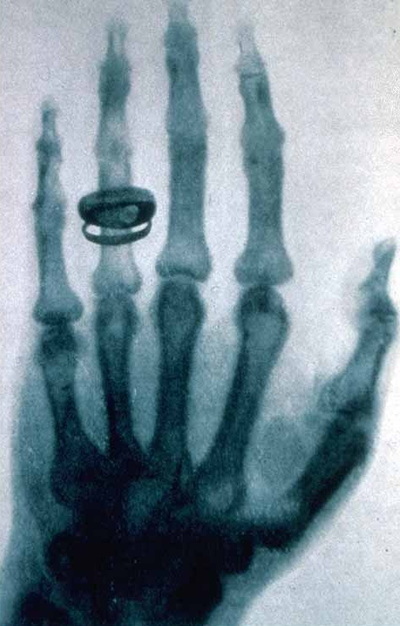
Discovery of X-Rays
On November 8, 1895, William Roentgen discovered X-rays during an experiment in which he passed an electric current through extremely low pressure gases, which created the X-ray beams. Aware of the dangers associated with prolonged exposure to the rays, Roentgen experimented extensively with his new discovery and despite using lead shields while working, he eventually died of intestinal carcinoma.
On November 8, 1895, William Roentgen discovered X-rays during an experiment in which he passed an electric current through extremely low pressure gases, which created the X-ray beams. Aware of the dangers associated with prolonged exposure to the rays, Roentgen experimented extensively with his new discovery and despite using lead shields while working, he eventually died of intestinal carcinoma.
Further experimentation with X-rays by Sir Joseph Larmor revealed in 1898 that accelerated particles do release radiation, confirming the authenticity of Roentgen's findings. As X-rays were used more frequently to research various aspects of the scientific world, Alfred-Marie Lienard, along with others, realized that by moving particles in a circular motion, the centripetal acceleration causes the particles to emit X-rays. In 1912, when attempting to navigate electromagnetic beams through a crystalline lattice, Max von Laue realized that the shorter wavelengths of X-rays could interfere with the structure and give details about composition. He was later awarded the Nobel Prize for his achievement in creating the first X-ray crystallography images. From this point, the field was of great interest to scientists seeking to open the door into the vast world of microscopic structures, and crystallography was the key.
What are macromolecules?
In biology and chemistry, macromolecules include four main groups: carbohydrates, proteins, nucleic acids, and lipids. The basic structures of these molecules are retained, but added functional groups can change the properties or behaviors of the overall molecule.
Carbohydrates
Carbohydrates include starch, glycogen, chitin, and cellulose, among others. The monomer (smallest unit) of carbohydrates is a carbon ring or chain with a variety of hydrogens and other organic compounds attached. For carbohydrates, the monomer is called a monosaccharide, which is more commonly known as simple sugars. Some well known examples of these monomers include glucose, fructose, and galactose, all of which have the ability to bond with other sugars through dehydration or condensation reactions, forming larger polysaccharides. This involves the removal of water so that the monomers can join together. Breaking apart the polymers, or polysaccharides in this case, involves hydrolysis, or the introduction of water, which splits the larger molecule.
Proteins
An important component to allow living things to move and aid in carrying out key chemical reactions. Structure is extremely important for the correct task being carried out by the protein, and slight changes can dramatically affect how the protein operates. Enzymes are a type of protein that lowers the activation energy for chemical reactions to occur by being in the correct conformation to facilitate the reactions. This forcing of the substrates (components of the reaction) together and providing the appropriate environment, the products can be made more effectively. Also, the enzyme is unchanged by the reaction, although it may change shape during due to the induced fit model. There are several levels to protein structure, which make them difficult to understand, yet vital when they are mastered. See the slideshow above for a visual understanding of the four levels of structure. X-ray crystallography is generally targeted at proteins more than the other macromolecules because they all have complex structure, crucial to their function. If this shape is altered or damaged, it is possible the molecule will no longer carry out its task. Oftentimes, this results in a disease or illness, which is why understanding the structure of proteins gives a great deal of insight into why a protein is malfunctioning and how it can be fixed.
Lipids
Lipids, also known as fatty acids, are hydrocarbons. This means that they are composed of a carbon chain with hydrogen atoms covalently bonded to the remaining free electrons. Variations of this basic structure are common because if one or more of the bonds between carbons were double or triple bonds, the chains would no longer be saturated. Also, if functional groups were to be added, the function of the larger molecule would be altered. A common and absolutely essential function of lipids is in the membrane of cells, because the phospholipid bilayer composes the majority of the cell membrane. Phospholipids are amphipathic, meaning that they have a hydrophobic tail, but a hydrophilic head so they orient themselves in a bilayer. This arrangement enables the selective permeability of the membrane, determining which compounds and materials are allowed to enter and leave the cell, requiring membrane proteins for the transport of certain molecules. While the lipid structure is well understood, the proteins embedded in this membrane are often the target of X-ray crystallography research. Some examples of this include the aquaporin, which is the channel allowing water to pass through the membrane, and the sodium-potassium pump, which is extremely important as a carrier-protein on the membrane.
Nucleic Acids
The monomer of nucleic acids is the nucleotide, a three-carbon, two-nitrogen base bonded to a ribose or deoxyribose sugar and a phosphate group. There are five main variations composing all the possible RNA and DNA bases. The multitude of possible combinations of these bases gives each DNA or RNA sequence its own structure and additional proteins found aid in coiling and twisting it into the final shape. The antiparallel and double helical structure of DNA was difficult to determine, so the use of X-ray crystallography allowed scientists to circumvent the minute size of molecules and understand the structure.
Why analyze macromolecules?
Macromolecules make up nearly the entire living world, so fully comprehending how they function is critical for further progress in medical applications, the development of nanotechnology, as well as crucial for additional steps towards complete knowledge.
In biology and chemistry, macromolecules include four main groups: carbohydrates, proteins, nucleic acids, and lipids. The basic structures of these molecules are retained, but added functional groups can change the properties or behaviors of the overall molecule.
Carbohydrates
Carbohydrates include starch, glycogen, chitin, and cellulose, among others. The monomer (smallest unit) of carbohydrates is a carbon ring or chain with a variety of hydrogens and other organic compounds attached. For carbohydrates, the monomer is called a monosaccharide, which is more commonly known as simple sugars. Some well known examples of these monomers include glucose, fructose, and galactose, all of which have the ability to bond with other sugars through dehydration or condensation reactions, forming larger polysaccharides. This involves the removal of water so that the monomers can join together. Breaking apart the polymers, or polysaccharides in this case, involves hydrolysis, or the introduction of water, which splits the larger molecule.
Proteins
An important component to allow living things to move and aid in carrying out key chemical reactions. Structure is extremely important for the correct task being carried out by the protein, and slight changes can dramatically affect how the protein operates. Enzymes are a type of protein that lowers the activation energy for chemical reactions to occur by being in the correct conformation to facilitate the reactions. This forcing of the substrates (components of the reaction) together and providing the appropriate environment, the products can be made more effectively. Also, the enzyme is unchanged by the reaction, although it may change shape during due to the induced fit model. There are several levels to protein structure, which make them difficult to understand, yet vital when they are mastered. See the slideshow above for a visual understanding of the four levels of structure. X-ray crystallography is generally targeted at proteins more than the other macromolecules because they all have complex structure, crucial to their function. If this shape is altered or damaged, it is possible the molecule will no longer carry out its task. Oftentimes, this results in a disease or illness, which is why understanding the structure of proteins gives a great deal of insight into why a protein is malfunctioning and how it can be fixed.
Lipids
Lipids, also known as fatty acids, are hydrocarbons. This means that they are composed of a carbon chain with hydrogen atoms covalently bonded to the remaining free electrons. Variations of this basic structure are common because if one or more of the bonds between carbons were double or triple bonds, the chains would no longer be saturated. Also, if functional groups were to be added, the function of the larger molecule would be altered. A common and absolutely essential function of lipids is in the membrane of cells, because the phospholipid bilayer composes the majority of the cell membrane. Phospholipids are amphipathic, meaning that they have a hydrophobic tail, but a hydrophilic head so they orient themselves in a bilayer. This arrangement enables the selective permeability of the membrane, determining which compounds and materials are allowed to enter and leave the cell, requiring membrane proteins for the transport of certain molecules. While the lipid structure is well understood, the proteins embedded in this membrane are often the target of X-ray crystallography research. Some examples of this include the aquaporin, which is the channel allowing water to pass through the membrane, and the sodium-potassium pump, which is extremely important as a carrier-protein on the membrane.
Nucleic Acids
The monomer of nucleic acids is the nucleotide, a three-carbon, two-nitrogen base bonded to a ribose or deoxyribose sugar and a phosphate group. There are five main variations composing all the possible RNA and DNA bases. The multitude of possible combinations of these bases gives each DNA or RNA sequence its own structure and additional proteins found aid in coiling and twisting it into the final shape. The antiparallel and double helical structure of DNA was difficult to determine, so the use of X-ray crystallography allowed scientists to circumvent the minute size of molecules and understand the structure.
Why analyze macromolecules?
Macromolecules make up nearly the entire living world, so fully comprehending how they function is critical for further progress in medical applications, the development of nanotechnology, as well as crucial for additional steps towards complete knowledge.